ViewSonic VB-PEN-002 Report - Page 2
Test Report, Report No.: GZAFN160301067X-1, Date: Apr 06 2016, TEST RESULTS
 |
View all ViewSonic VB-PEN-002 manuals
Add to My Manuals
Save this manual to your list of manuals |
Page 2 highlights
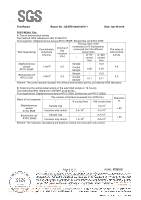
Test Report Report No.: GZAFN160301067X-1 Date: Apr 06 2016 TEST RESULT(S): A: Test of antimicrobial activity Test method: With reference to ISO 22196:2011 Test organism: Staphylococcus aureus ATCC 6538P, Escherichia coli ATCC 8739 Test Organism(s) Concentration of bacteria (cfu/mL) Volume of test inoculum (mL) The log value of the number(cfu/cm2) that bacteria recovered from the different contact time at "0h" at "24h" / contact contact time time The value of antimicrobial activity Staphylococcus Sample / -0.21 aureus ATCC 6538P 1.1х106 0.2 Control Sample 4.08 4.58 4.8 Escherichia coli ATCC 8739 1.0х106 Sample / -0.21 0.2 Control Sample 4.11 5.11 5.3 Remark: The control sample is plastic film without antimicrobial activity, provided by SGS laboratory. B: Determine the antimicrobial activity of the submitted sample in 18 hour(s) Test method(s):With reference to ASTM E 2149-2013a Test organism(s): Staphylococcus aureus ATCC 6538, Escherichia coli ATCC 25922 Name of test organism The number of bacteria recovered from (CFU/mL) / "0"contact time "18h"contact time Staphylococcus aureus ATCC 6538 Escherichia coli ATCC 25922 Sample (1g) Inoculum only control Sample (1g) Inoculum only control / 2.4×105 / 1.7×105 1.9×101 4.2×105 2.2×101 2.5×104 Remark: The reduction calculations are based on values from inoculum only control. Reduction % > 99 > 99 Page 2 of 3 RAND: 3750310