HP 35s HP 35s scientific calculator - User Guide - Page 127
Stores 297.1
 |
UPC - 000000000000
View all HP 35s manuals
Add to My Manuals
Save this manual to your list of manuals |
Page 127 highlights
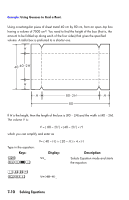
A 2-liter bottle contains 0.005 moles of carbon dioxide gas at 24°C. Assuming that the gas behaves as an ideal gas, calculate its pressure. Since Equation mode is turned on and the desired equation is already in the display, you can start solving for P: Keys: Display: Description: P value value value value Solves for P; prompts for V. Stores 2 in V; prompts for N. Stores .005 in N; prompts for R. Stores .0821 in R; prompts for T. Calculates T (Kelvins). Stores 297.1 in T; solves for P in atmospheres. A 5-liter flask contains nitrogen gas. The pressure is 0.05 atmospheres when the temperature is 18°C. Calculate the density of the gas (N × 28/V, where 28 is the molecular weight of nitrogen). Keys: Display: Description: Displays the equation. Solves for N; prompts for P. Stores .05 in P; prompts for V. Stores 5 in V; prompts for R. Retains previous R; prompts for T. Calculates T (Kelvins). Solving Equations 7-5